Key Words
Combustion: rapid chemical combination, or burning, of a substance with oxygen producing heat and light
Decay: the process by which a metal combines with oxygen causing rotting or decomposition
Paramagnetic: contains unpaired electrons
Decay: the process by which a metal combines with oxygen causing rotting or decomposition
Paramagnetic: contains unpaired electrons
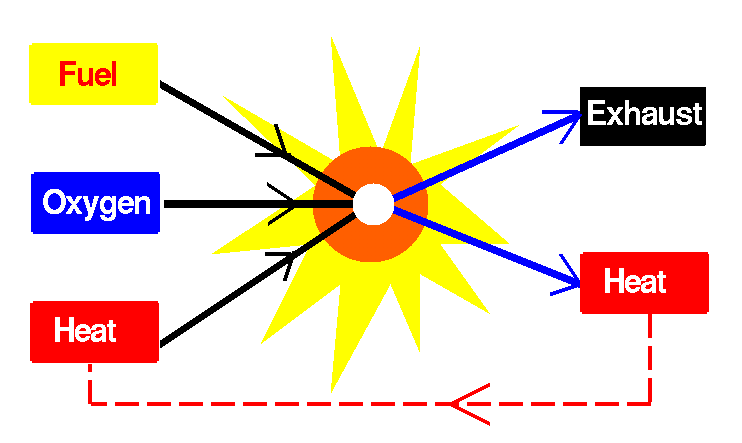
Chemical Properties Oxygen is a highly reactive element, highly paramagnetic, and is easily capable of combining with other elements. One of oxygen's most important chemical properties is that it supports combustion (see picture to the right). Oxygen also combines with elements at room temperature, for example, the formation of rust. Decaying is an example of oxygen reacting with compounds. Carbon dioxide and water are the main products of decay.
|
Process of Combustion: Fuel + Oxygen + A Source of Heat
|