Key Words
Allotropes: forms of an element with different physical and chemical properties
|
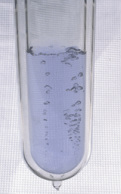
Liquid oxygen in an unsilvered flask. Oxygen in the liquid state is a pale blue substance.
The structure shown above is oxygen's cubic crystal element structure.
|
Physical Properties Oxygen exists in all three forms - liquid, solid, and gas. The liquid and solid forms are a pale blue colour. However, oxygen gas is colourless, odourless, and tasteless. The elemental structure is a cubic crystal shape.
Oxygen changes from a gas to a liquid at a temperature of 182.96°C, and then can be solidified or frozen at a temperature of -218.4°C. Oxygen exists in all three allotropic forms. The three allotropic forms include normal oxygen, diatomic oxygen, or dioxygen; nascent, atomic, or monatomic oxygen; and ozone or triatomic oxygen. The three allotropes differ in several ways; such as, atoms and molecules. For example, the oxygen we're most familiar with in the atmosphere has two atoms in every molecule. Nascent oxygen only has one atom in every molecule, and the third allotrope (ozone) has three atoms in every molecule. |